GLYCOGEN METABOLISM
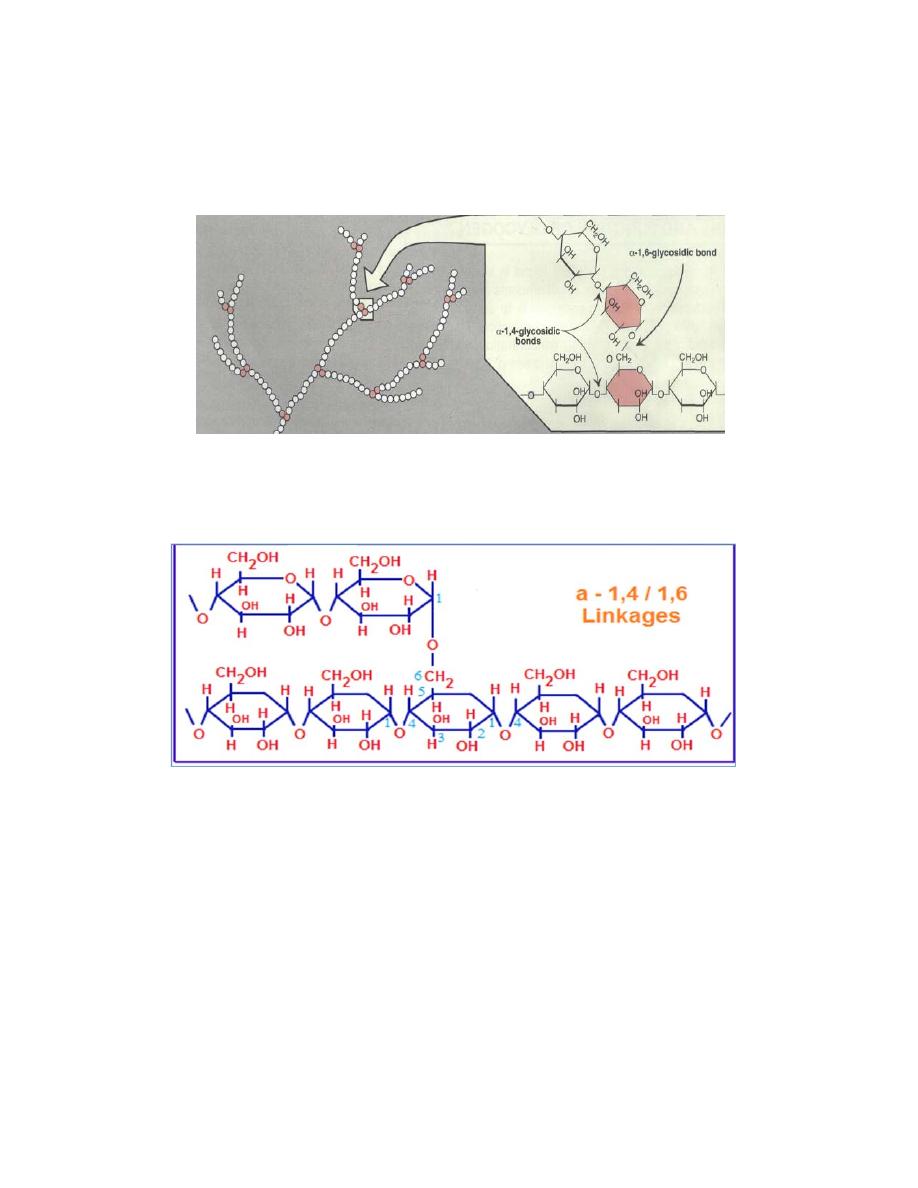
Glycogen is a branched homopolysaccharides made exclusively from a-
D-glucose.
The primary glycosidic bond is an alpha-1,4 glycosidic linkage. After an
average of eight to ten glucosyl residues (glucose), there is a branch
containing an alpha-1,6 glycosidic linkage .
Glycogen is the storage form of carbohydrates in the human body. The
major sites of storage are liver and muscle. The major function of liver
glycogen is to provide glucose during fasting. when blood glucose level
falls, liver glycogen is broken down and helps to maintain blood glucose
level. After taking food, blood glucose tends to rise, which causes
glycogen deposition in liver. About 5 hours after taking food, the blood
glucose tends to fall, but, glycogen is lysed to glucose so that the energy
needs are met. After about 18 hours fasting, most of the liver glycogen is
depleted, at this time depot fats are hydrolysed and energy requirement is
met by fatty acid oxidation.The function of muscle glycogen is to act as
reserve fuel for muscle contraction.

DEGRADATION OF GLYCOGEN (GLYCOGENOLYSIS
)
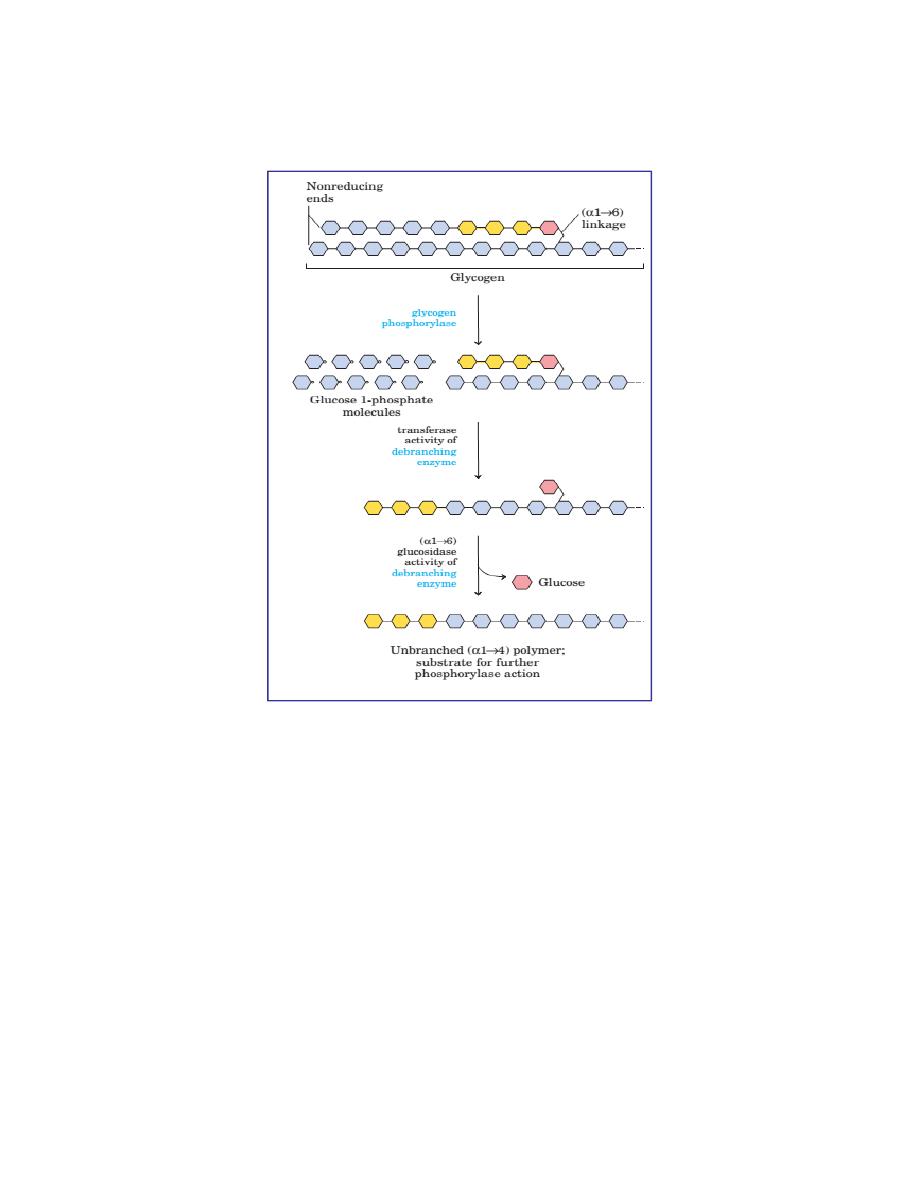
1. Glycogen Phosphorylase
Glycogen phosphorylase removes glucose as glucose-1-phosphate from
glycogen. It removes glucose units one at a time. Enzyme sequentially
hydrolyses alpha-1,4 glycosidic linkages, till it reaches a glucose residue,
3-4 glucose units away from a branch point. It cannot attack the 1,6
linkage at branch point. If glycogen phosphorylase alone acts on a
glycogen molecule, the final product is a highly branched molecule; it is
called limit dextrin. PLP (pyridoxal phosphate) is an essential cofactor in
the glycogen phosphorylase reaction.
2. Debranching by bifunctional two Enzymes
Then a block of 3 glucose residues (trisaccharide unit) are transferred
from the branching point to another branch by enzyme alpha-1,4 →
alpha-1
,
4 glucan transferase. Now the branch point is free. Then alpha-
1,6- glucosidase (debranching enzyme) can hydrolyse the remaining
glucosyl unit held in alpha-1,6 linkage at the branch point. This glucose
residue is released as free glucose. At this stage, the ratio of glucose-1-
phosphate to free glucose is about 8:1.The transferase and alpha-1,6-
glucosidase will together convert the branch point to a linear one. With
the removal of the branch point, then phosphorylase enzyme can proceed
with its action.
3. Phosphoglucomutase
Phosphorylase reaction produces glucose-1- phosphate while debranching
enzyme releases glucose. The glucose-1-phosphate is converted to
glucose-6-phosphate by phosphoglucomutase
4. Glucose-6-phosphatase in Liver
Next, hepatic glucose-6-phosphatase hydrolyses glucose-6-phosphate to
glucose. The free glucose is released to the blood stream.
5. Muscle Lacks Glucose-6-phosphatase
Muscle will not release glucose to the blood stream, because muscle
tissue does not contain glucose-6-phosphatase. Instead, in muscle,
glucose-6-phosphate undergoes glycolysis to produce ATP for muscle
contraction.
GLYCOGEN SYNTHESIS (GLYCOGENESIS)
Glycogen is synthesized from molecules of a-D-glucose. The process
occurs in the cytosol. Glycogen synthesis takes place in virtually all
animal tissues but is especially prominent in the liver and skeletal
muscles.
The
starting
point
for
synthesis
of
glycogen
is glucose 6-phosphate. This can be derived from free glucose in a
reaction catalyzed by hexokinase (glucokinase) ,To initiate glycogen
synthesis, the glucose 6-phosphate is converted to glucose 1-phosphate.
The glycogen synthesis occurs by a pathway different from the reversal
of glycogenolysis. The steps are:
1. Activation of Glucose
UDP glucose is formed from glucose-1-phosphate and UTP (uridine
triphosphate) by the enzyme glucose-1-uridyltransferase.
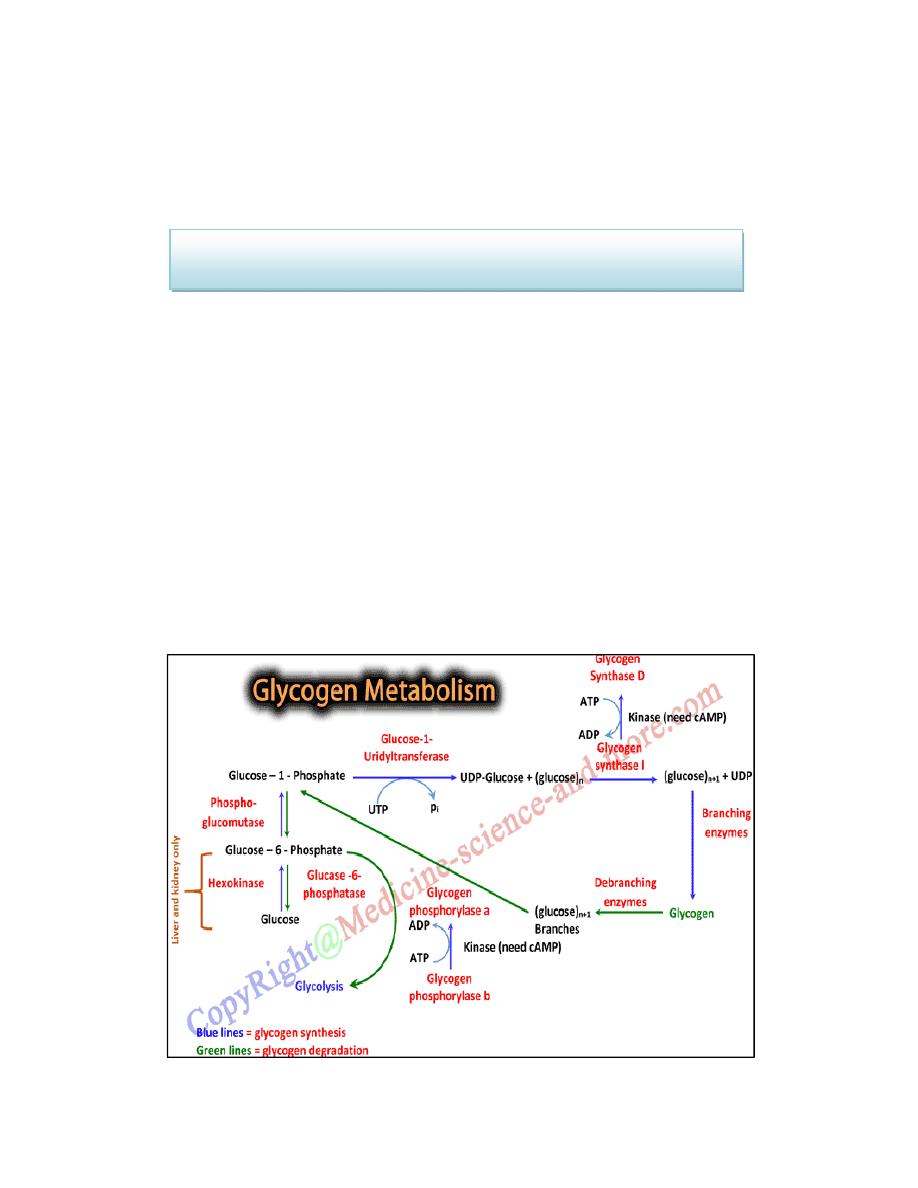
2. Glycogen Synthase
The glucose moiety from UDP-glucose is transferred to glycogenin (a
glycogen primer molecule which is essential to accept the glycosyl unit),
The primer is made up of a protein-carbohydrate complex.
Glycogen synthase
Glycogen primer (n) + UDP glucose ————→ Glycogen (n+1) + UDP
In this step, activated glucose units are sequentially added by the enzyme
glycogen synthase .The glucose unit is added to the non-reducing (outer)
end of the glycogen primer to form an alpha-1,4 glycosidic linkage and
UDP is liberated.
3. Branching Enzyme
The glycogen synthase can add glucose units only in alpha-1,4 linkage. A
branching enzyme is needed to create the alpha-1,6 linkages. When the
chain is lengthened to 11 – 12 glucose residues, the branching enzyme
will transfer a block of 6 to 8 glucose residues from this chain to another
site on the growing molecule. The enzyme amylo-[1,4]→[1,6]-
transglucosidase (branching enzyme) forms this alpha-1,6 linkage . To
this newly created branch, further glucose units can be added in alpha-1,4
linkage by glycogen synthase.

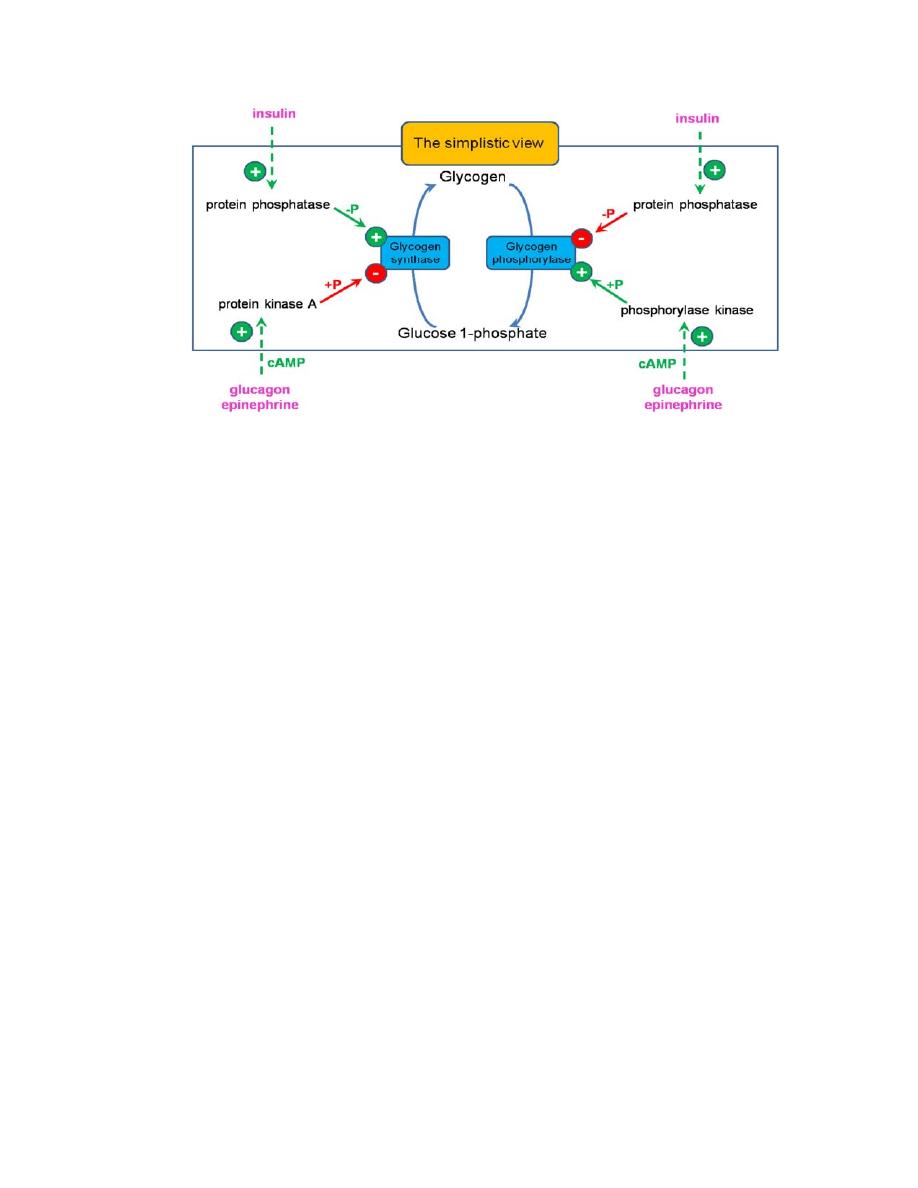
Regulation of Glycogen Metabolism
Glycogen metabolism is regulated by coordinated regulation of glycogen
synthase and glycogen phosphorylase. The regulatory mechanisms
include:
Allosteric regulation of glycogen synthase and phosphorylase
The allosteric activators and inhibitors of phosphorylase and synthase are
listed below.
Covalent modification (phosphorylation and dephosphorylation)
of enzymes by hormonal control
Glycogenolysis and glycogenesis pathways are reciprocally regulated to
prevent futile cycles.
Glycogen phosphorylase is activated by phosphorylation by kinase that
adds phosphate group to a specific serine residue of phosphorylase while
phosphorylase inactivated by dephosphorylation by phosphatase .The
active form of phosphorylase is referred to as ‘a’ (active, phosphorylated)
and the relatively inactive dephosphorylated form as ‘b’.
The same protein kinase, which phosphorylates the phosphorylase, would
also phosphorylate glycogen synthase. The activity of the glycogen
synthase is markedly decreased on phosphorylation; Glycogen synthase is
active in the dephosphorylated state. So, the active glycogen synthase (a)
is dephosphorylated and phosphorylated (b) is relatively inactive.
Insulin and glucagon are the major regulatory hormones, although
epinephrine has stimulatory effect on glycogenolysis in both liver and
muscle. Insulin promotes glycogen synthesis in muscle and
liver by favoring dephosphorylation of enzymes. Glycogen phosphorylase
is activated in response to glucagon or epinephrine, which converts
glycogen phosphorylase (b) to its active (a) form .
GLYCOGEN STORAGE DISEASES
Inherited deficiencies in specific enzymes of glycogen metabolism in
both liver and muscle are the causes of glycogen storage diseases. These
are inborn-errors of metabolism.
Glycogen Storage Disease Type-I
It is also called Von Gierke's Disease. Most common type of glycogen
storage disease is type I. Incidence is 1 in 100,000 live births. In this
disease Glucose-6-phosphatase is deficient, fasting hypoglycemia that
does not respond to stimulation by adrenaline. The glucose cannot be
released from liver during overnight fasting, Hyperlipidemia, lactic
acidosis and ketosis. Glycogen gets deposited in liver. Massive liver
enlargement may lead to cirrhosis. Children usually die in early
childhood. Treatment is to give small quantity of food at frequent
intervals. Other glycogen storage diseases (type II to X); they are very
rare, incidence being 1 in 1 million births.
